Plenity®

Stimulant-free and non-habit forming
Clinically tested and FDA-cleared
How Plenity worked
Plenity capsules absorb the water you drink mixed with the food you eat to make you feel full and help you eat less.
Take 3 capsules
Take 3 Plenity capsules before lunch and again before dinner.
Drink 16 oz of water
Don’t just take a sip—for the capsules to take effect, you need to drink 16 oz of water with each dose.
Wait 20 minutes
You’re ready to enjoy your meal and feel full after eating smaller portions.
Effective
Nearly 60% of study participants that were treated with Plenity achieved an average weight loss of 10% of their body weight or ~22 pounds.
Proven safety profile
A clinical study showed that Plenity is as safe and tolerable as a placebo (a capsule with inactive ingredients), with minor side effects.
Non-stimulant
Plenity is not a stimulant and there are no restrictions for how long it can be used.
Accessible
Plenity is safe for use by people with or without other conditions such as hypertension, type 2 diabetes, or dyslipidemia.
Check out Ro’s weight loss offering
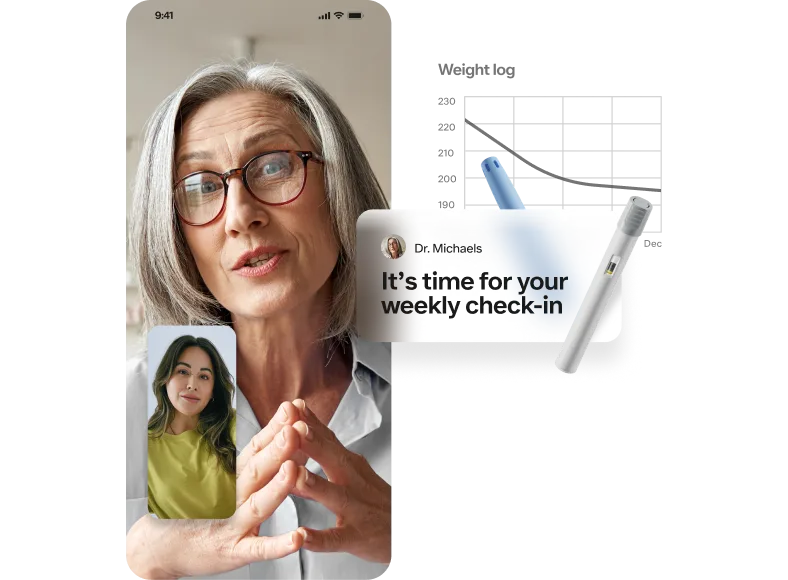
The Body Program pairs GLP-1 medications with healthy lifestyle changes to help you achieve sustainable weight loss.
Important safety information
What you should know before taking Plenity