Insulin's discovery and its role in the body

Reviewed by Tzvi Doron, DO,
Written by Mike Bohl, MD, MPH, ALM
last updated: Aug 28, 2019
9 min read
Here's what we'll cover
On a rainy night in 1921 at the University of Toronto, Frederick Banting, along with his assistant Charles Best, discovered insulin in a laboratory provided to them by John Macleod. In so doing, they achieved one of the most significant medical breakthroughs of all time.
Ok, we don’t know if it was actually raining. And the breakthrough certainly didn’t occur in just one night. But these three scientists - along with James Collip, who later helped purify insulin - are indeed credited with the discovery of insulin.
The true story of insulin’s discovery spans several years and a legion of researchers (Vecchio, 2018). While descriptions of a disease assumed to be diabetes date as far back as 1500 BCE in ancient Egypt, it wasn’t until the 1800s that people began realizing the pancreas may play a role in the disease. In 1890, researchers noticed that complete removal of the pancreas from a dog resulted in the symptoms often seen in people with diabetes. From this, they knew the pancreas had something to do with the condition—they just had to figure out what precisely that was.
The pancreas is a unique organ that has both exocrine and endocrine functions. “Exocrine” means an organ secretes chemical substances through ducts. In this case, the pancreas secretes digestive enzymes through ducts and into the intestines. “Endocrine” means an organ secretes chemical substances directly into the bloodstream.
Researchers noticed that the pancreas had multiple functions. When they blocked the ducts of the pancreas (its exocrine function), parts of the pancreas would die. However, areas of the pancreas known as the islets of Langerhans (because they looked like islands of tissue in the pancreas) did not die, and blood sugar was not affected. This indicated that the islets had some sort of endocrine function that played a role in blood sugar levels. In the 1910s, Sir Edward Sharpey-Schafer coined the term “insuline” to refer to the substance secreted from these islets that could control blood sugar, taking the term from the Latin “insula,” meaning “island.”
Banting and Best worked on isolating insulin from the pancreas in 1921 by conducting research on dogs. They presented their findings—officially announcing the discovery of insulin—in December 1921 and in 1922, insulin was first used on humans. The first insulin recipient was Leonard Thompson, a 14-year-old boy with type 1 diabetes. Before this time, type 1 diabetes mellitus had been a fatal disease.
Weight loss
Get access to GLP-1 medication (if prescribed) and 1:1 support to meet your weight goals
What is insulin, and why is it so important?
As Banting and others discovered, insulin is a hormone that is secreted from areas of the pancreas called the islets of Langerhans. Specifically, insulin is secreted from beta cells in the islets. There are also alpha cells that secrete glucagon (another hormone involved in regulating blood sugar levels) delta cells that secrete somatostatin (an inhibiting hormone), and smaller amounts of other hormone-secreting cells.
Insulin and glucagon are the primary hormones that regulate blood sugar levels in the body. To stay healthy, it is essential for blood sugar levels to remain tightly controlled. Having blood sugar levels that are too low (called hypoglycemia) or blood sugar levels that are too high (hyperglycemia) can lead to complications and even death. Insulin helps lower blood sugar levels while glucagon helps raise them. Disorders of insulin are, therefore disorders in which blood sugar levels are uncontrolled. This primarily refers to diseases like type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM). However, some uncommon conditions can impact blood insulin levels as well. We’ll get to those soon, but first, let’s take a deeper dive into what insulin does in the body.
Insulin in the body
Insulin is a type of hormone known as a peptide hormone. Peptide hormones work in the body by acting on the surface of cells to pass along messages. Insulin starts as a single chain of amino acids known as preproinsulin before it is converted into proinsulin, and then finally into insulin. Part of this process creates a byproduct known as the C-peptide. Measuring the C-peptide can be important clinically because it can give an indication of how much natural insulin a person’s body is producing.
Insulin is released from the beta cells into the bloodstream in response to elevated blood sugar levels (also called blood glucose levels). When you eat food that has sugar or carbohydrates (which get broken down into sugar), blood glucose levels go up. Glucose molecules enter the beta cells in the pancreas by going through a protein on the cellular surface called glucose transporter 2 (GLUT2). This signals to the beta cell that it is time to secrete insulin.
Once released into the body, insulin binds to insulin receptors on the surface of cells. In fat (adipose) cells and muscle cells, activation of the insulin receptor moves a protein called glucose transporter 4 (GLUT4) to the surface of the cells. Glucose is then able to enter into fat and muscle cells through GLUT4, effectively lowering the amount of glucose in the blood. In fat cells, glucose is stored as triglycerides (a type of fat). In muscle cells, glucose is used for energy and can also be stored for later in long chains called glycogen.
Insulin also acts on liver cells, prompting them to store glucose as glycogen as well. Glucose can enter liver cells without GLUT4. However, insulin activates an enzyme called hexokinase and other enzymes that are involved in making glycogen, effectively trapping the glucose there and keeping blood sugar levels down.
It is possible to do a blood test to check levels of insulin in the body. However, these values are not routinely checked. Instead, other blood tests can be used to diagnose diseases like prediabetes, T1DM, and T2DM, including the fasting blood glucose (FPG) test, the oral glucose tolerance test (OGTT), and the hemoglobin A1c (HbA1c) test.
Disorders of insulin
Disorders of insulin include states in which there is too much insulin in the body (called hyperinsulinemia), states in which there is not enough insulin in the body, and states in which there is a variable amount of insulin, but the body’s cells are not responding to it effectively. The latter situation is called insulin resistance and is the cause of prediabetes and T2DM.
Prediabetes, T1DM, and T2DM are the most common disorders of insulin by far. According to the Centers for Disease Control and Prevention (CDC), as of 2015, over 84 million Americans have prediabetes - a condition that can lead to T2DM (CDC, 2017). Additionally, over 30 million—that’s nearly 1 in 10—people in the United States have diabetes. Of those, 90-95% of people have T2DM.
T1DM is an autoimmune disease in which the body stops making insulin or only makes very little insulin. An autoimmune disease is a disorder in which the body attacks itself. In T1DM, the body makes antibodies that target and destroy the beta cells of the pancreas. As a result, they are no longer able to produce insulin. T1DM used to be referred to as juvenile diabetes or juvenile-onset diabetes, however, it is possible to be newly diagnosed with T1DM as an adult. Genetics appears to play at least a partial role in putting somebody at risk of developing T1DM, but it is not the only factor. Researchers believe that exposures, such as exposures to viruses, also play a role. Because people with T1DM do not make insulin, they are dependent on insulin as a treatment.
T2DM and prediabetes are diseases in which the body is resistant to insulin. This means that the pancreas is still able to make insulin (at least, at first) but the body’s cells have decreased insulin sensitivity. This leads to rising blood sugar levels because glucose is not effectively transported into fat and muscle cells, nor is it effectively kept in the liver as glycogen. When levels are mildly elevated, an individual is said to have prediabetes. Once blood sugar levels go above a certain threshold, an individual is diagnosed with T2DM.
Researchers aren’t sure exactly what causes insulin resistance on a molecular level, but the most significant risk factors are living a sedentary lifestyle and being overweight or obese (particularly having excess fat around the middle or an increased waist size). At first, the beta cells of the pancreas may be able to “keep up” with the body’s needs, compensating for insulin resistance. However, over time, the pancreas gets “burned out” and cannot make as much insulin as the body needs. Medications for T2DM are geared towards decreasing blood sugar levels and making the body more sensitive to insulin. A little under a third (31%) of people with T2DM require insulin as a treatment.
There are a few rarer disorders that impact insulin levels in the body. Insulinomas are tumors that secrete insulin, resulting in high blood insulin levels and low blood sugar levels. These are exceedingly rare, with an estimated 4 cases per 1,000,000 people each year (UpToDate, 2019). Nesidioblastosis is another condition in which the body produces too much insulin. However, instead of having an insulin-secreting tumor, people with nesidioblastosis just have a higher number of beta cells in the pancreas than normal. Lastly, there are genetic causes of high blood insulin that some individuals are born with. This is called congenital hyperinsulinism. So far, nine gene mutations have been identified that can cause this, but they too, are very rare—1 in 2,500 to 1 in 50,000.
What are the signs and symptoms of insulin problems?
The symptoms associated with insulin problems are the same as the symptoms that present when blood sugar levels are too high or too low.
When insulin levels are too high, it can cause hypoglycemia. Symptoms of low blood sugar may start out mild but can rapidly become dangerous and even fatal.
They include:
Dizziness
Fatigue
Headache
Irregular heartbeat or palpitations (fast heartbeat that feels like a fluttering)
Shakiness
Sweating
Changes in vision
Confusion
Slurring of words
Seizures
Loss of consciousness
Death
When insulin levels are too low, or the body is resistant to insulin, it can cause hyperglycemia. High blood sugar may be asymptomatic, may result in long-term complications, or may result in life-threatening conditions known as diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS).
Symptoms include:
Increased thirst (polydipsia)
Frequent urination (polyuria)
Extreme hunger (polyphagia)
Lack of energy
Weight loss
Vision changes
Numbness, tingling, or pain in the extremities (neuropathy)
Difficulty healing
Frequent infections
Heart disease or blood vessel problems
Fruity breath
Drowsiness
Abdominal pain
Vomiting
Fever
Confusion
Coma
Insulin as a medication
Ever since its discovery, insulin has been further tested and developed into several medicines that can be given to people with diabetes. The American Diabetes Association (ADA) estimates that six million people use insulin in the United States (ADA, 2015).
Unlike other diabetes medications, insulin is not a pill. With one exception, it must be given through the skin, usually with a needle. That being said, there are multiple options for people who are on insulin (Shah, 2016).
Vial and syringe: The vial and syringe method is what you may typically think of when you envision insulin injections or insulin shots. It involves the user drawing up a predetermined dose of insulin into a syringe, which they then use to inject themselves.
Insulin pen: Insulin pens are devices that already have insulin inside of them. The user can adjust the dose, typically with a dial or some other mechanism. Insulin pens are more user-friendly and may also have smaller needles, making them more comfortable to use.
Insulin pump: Insulin pumps are devices that attach to the body that can deliver small amounts of insulin continuously as well as larger doses during mealtime. These pumps more closely mimic what the pancreas actually does in the body. They can also deliver more precise doses of insulin. Insulin pumps may allow an individual greater flexibility in their lifestyle since people only need to wear the device and do not have to worry about individual injections constantly.
When combined with continuous glucose monitoring, insulin pumps may serve as a closed-loop form of insulin delivery. This means that blood sugars levels are continuously monitored and the pump responds appropriately to the readings. This system is sometimes referred to as an artificial pancreas and is currently being tested out.
Insulin jet injector: Jet injectors work by shooting high-pressure liquid medication through the skin. They can be an effective delivery method for those who are uncomfortable with needles. One study has even demonstrated that the jet injector can be superior to the insulin pen in controlling blood sugar levels of people with T2DM (Guo, 2017).
Inhaled insulin: There is currently one form of FDA-approved inhaled insulin available called Afrezza. Afrezza is a rapid-acting form of insulin that reduces the number of daily needle sticks an individual may need. Other types of inhaled insulin used to be available but were not very popular, possibly due to respiratory side effects (Oleck, 2016).
Just like insulin delivery methods vary, so do the types of insulin people can give themselves. At first, insulin used for treatment was derived from animals, specifically cows (bovine insulin) and pigs (porcine insulin). Now, synthetic human insulin is made using bacteria. To make this happen, scientists used recombinant DNA technology. This entails taking the human gene for insulin production and place it into the genetic code of bacteria. The bacteria read the code as if it were their own and produce human insulin.
One of the goals of insulin treatment is to mimic what happens with insulin in the body. This includes a steady supply of low-level insulin that lasts throughout the day (this is called the basal insulin) coupled with increases in insulin around mealtimes to handle related increases in blood sugar (these are called boluses). With this in mind, several different forms of insulin have been developed that differ based on their time of onset (how long the insulin takes to start working), peak time (how long after injection the insulin is most effective), and duration (how long the effects of the insulin last for). These different forms are sometimes referred to as insulin analogs. Sorted by how quickly they work, the main types of insulin according to the ADA are:
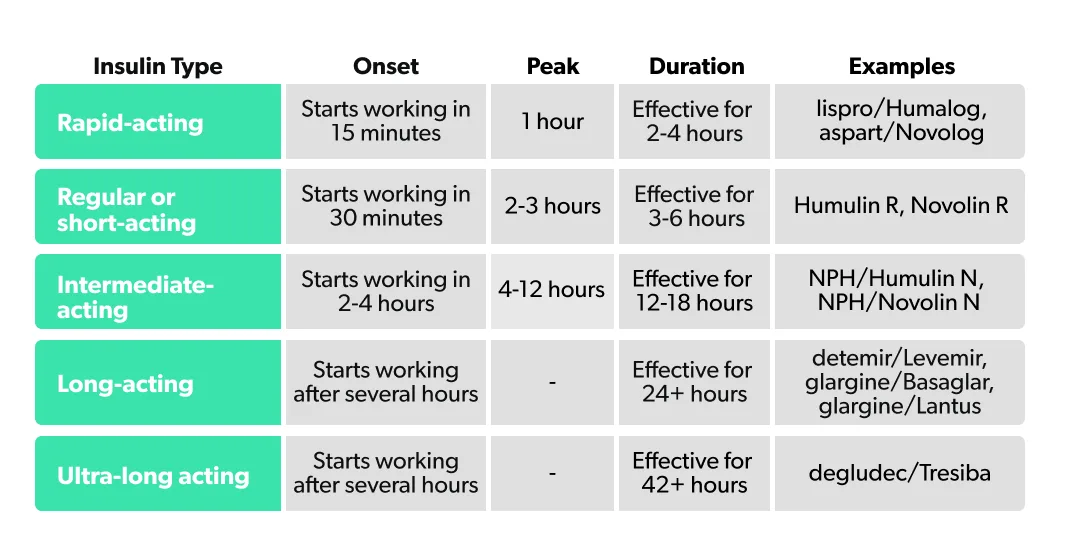
In some cases, different types of insulin may be combined.
If you are taking insulin for diabetes treatment, it is important you speak to a healthcare professional about your dosage. Keeping track of your blood glucose levels is a good way to identify if you are experiencing many episodes of hypoglycemia or hyperglycemia, which may indicate that your dosage should change.
DISCLAIMER
If you have any medical questions or concerns, please talk to your healthcare provider. The articles on Health Guide are underpinned by peer-reviewed research and information drawn from medical societies and governmental agencies. However, they are not a substitute for professional medical advice, diagnosis, or treatment.
American Diabetes Association. (n.d.). Insulin Basics. Retrieved from https://www.diabetes.org/diabetes/medication-management/insulin-other-injectables/insulin-basics
American Diabetes Association. (2015). Fast Facts Data and Statistics about Diabetes. Retrieved from https://professional.diabetes.org/content/fast-facts-data-and-statistics-about-diabetes
Centers for Disease Control and Prevention. (2017, July 18). New CDC report: More than 100 million Americans have diabetes or prediabetes. Retrieved from https://www.cdc.gov/media/releases/2017/p0718-diabetes-report.html
Guo, L., Xiao, X., Sun, X., & Qi, C. (2017). Comparison of jet injector and insulin pen in controlling plasma glucose and insulin concentrations in type 2 diabetic patients. Medicine , 96 (1). doi: 10.1097/md.0000000000005482. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/28072690
Oleck, J., Kassam, S., & Goldman, J. D. (2016). Commentary: Why Was Inhaled Insulin a Failure in the Market? Diabetes Spectrum , 29 (3), 180–184. doi: 10.2337/diaspect.29.3.180. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5001220/
Shah, R., Shah, V., Patel, M., & Maahs, D. (2016). Insulin delivery methods: Past, present and future. International Journal of Pharmaceutical Investigation , 6 (1), 1. doi: 10.4103/2230-973x.176456. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4787057/
UpToDate. (2019). Insulinoma. Retrieved from https://www.uptodate.com/contents/insulinoma
Vecchio, I., Tornali, C., Bragazzi, N. L., & Martini, M. (2018). The Discovery of Insulin: An Important Milestone in the History of Medicine. Frontiers in Endocrinology , 9 . doi: 10.3389/fendo.2018.00613. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/30405529









