Finasteride 1 mg
Hair loss treatment plan
Be sure to take your time and read everything below. It is essential for you to understand the potential risks and benefits of treatment. Note that shedding fine hairs is an expected early effect of finasteride (see below under Finasteride FAQs for more information). Please do not hesitate to reach out to our medical support team if you have ANY questions.
Welcome. Losing hair marks a change; whether it is embraced by taking a razor to a receding hairline or fought by taking medication to regrow what has been lost, the decision is never made without careful consideration. You may have felt like losing hair would be a losing battle and resigned yourself to the inevitable bald head that each of your uncles (and maybe an aunt or two) brandished at every family gathering. You may have silently resented that you were losing the appearance of youth while you still felt young and robust.
No matter what brought you here today, the fact is that you made a decision and took action. You have sought the advice and care of doctors. Your doctor, who reviewed your medical information, has formulated a plan. What you will read today is a plan that details how you can work to preserve hair or even regrow hair.
Medication works for the vast majority of patients and it usually does so without causing side effects—and that’s great. However, if a trusting relationship is to work, then it must be open, meaning that we must share more than how everything can go according to plan.
Sometimes, the plan doesn’t work. It is part of the reality that no drug is a magic bullet. And, just as no drug works on everyone, no drug is 100% safe. It is important you learn all you can. The more you read, the more questions will be answered and the better the plan can work.
Telemedicine has the advantage of convenience, but it relies on your honesty and involvement in the process. That includes reading everything below carefully (including the package insert, now online, and when it arrives in print with your medication), and communicating with your doctor. If your health should change, should you have a side effect, should the medicine not work or stop working over time, should you be prescribed ANY new medication or change your medication regimen, should you visit another doctor, please contact us.
Keep EVERY healthcare provider informed.
We are here for you. You’ve taken a big step and you are not alone. Read, learn, ask us questions. We have done the legwork and put together information so you can do it right.
First, you read your plan, which is followed by information about hair loss. Lastly, read the detailed information about the medication prescribed by your doctor. Read it all, and read the package insert and the official prescribers’ digital reference (PDR).
Your physician has reviewed your medical information and has prescribed finasteride to treat adrogenic alopecia. This medication helps patients maintain the hair they have or regrow some hair they have lost. The medications your physician has prescribed or asked you to use should only be used as directed.
Your physician has prescribed finasteride 1 mg tablets.
Be aware that finasteride can cause side effects and you should familiarize yourself with the list included in the section that discusses side effects. The vast majority of men take finasteride without difficulty, but familiarity with the possible side effects will help you identify if you are one of the men who experiences them.
Being fully informed is the only way for you to know if this plan suits your needs and if you want to accept it as presented. You may reject it or you may want to request a modification to the plan. Please, do not hesitate to make your voice heard. Contact us with any questions. We want the best for you and the only way to do that is if you are fully informed and if we work as a team.
Let’s begin. Read everything and keep all the literature handy (digitally, as well) in case your medical status changes and you need to refer back to it.
How should I take my finasteride?
It is best to take it each day at the same time. If you miss a dose, skip the missed dose and continue on your regimen the next day. Of course, if you are off by just a few hours, take your medication and get back onto your daily rhythm. It is important to take your medication without days of interruption, so be sure to pack enough when traveling, on vacation, etc. Finasteride can be taken with or without food.
How should I store my finasteride?
The pills should be kept dry and at room temperature between 68˚F to 77˚F (20˚C to 25˚C). The bathroom is a moist environment, so choose another place. They must be stored away from children and pets. Also, broken pills can expose the skin to finasteride. This is category X, meaning that it can cause birth defects. Though they are coated and generally considered safe when unbroken, women should never touch these pills.
How soon might I see results?
Finasteride takes 3 to 4 months to show any benefit. It can take up to a year to see the maximum results. Moreover, as finasteride enters the system, it may even cause more shedding of fine hairs. Do not be concerned – this is expected. Also, some men do not respond to the medication; this can be as high as 1 out of every 10 men.
As long as you do not experience side effects, it is best to continue medication for one full year before deciding whether the medication is doing all it can for you.
What is finasteride and how does it work?
Finasteride blocks the conversion of testosterone to dihydrotestosterone (DHT). Male pattern baldness (MPB) or androgenic alopecia, is caused not by testosterone, but by DHT. It is DHT that causes hair follicles to shrink and lose the ability to create new hairs. This is called “miniaturization.”
Testosterone is changed into DHT by an enzyme called 5 alpha-reductase. That enzyme comes in different forms and one form of the enzyme converts testosterone into DHT in the prostate and on the scalp.
Researchers first tried treating enlarged prostates with finasteride. Finasteride is called a 5 alpha-reductase inhibitor, meaning it stops the enzyme from changing testosterone into DHT. They ended up finding that the medication stopped hair loss in the study patients and even regrew hair for some.
It is unknown why the hair follicles of some men are more predisposed to DHT – causing follicles to shrink – but genetics is a big part of it. No one knows precisely how a man’s genes cause hair loss, but there are over 250 genes that can come from either parent.
It is a man’s genes that determine if he will lose his hair or not. The genes could code for more receptors (places for DHT to “land”) on a hair follicle, or make them more sensitive to DHT. That being said, we do know that blocking DHT can slow or even reverse hair loss.
In one European study (Euro J of Dermatol 2008; 18: 400–6), men treated with 1 mg finasteride over a five year period “led to a 93% decrease relative to placebo in the 5-year likelihood of developing further hair loss.” That is excellent and has been reproduced in other studies—but does it regrow hair?
In one analysis, “Three double-blind, randomized, placebo-controlled Phase III studies of finasteride were conducted in 1879 men ages 18–41 years with mild to moderate AGA (this stands for Androgenic Alopecia, which is male pattern baldness).”
“Two of the studies enrolled men with predominantly vertex hair loss …and one study enrolled men with predominantly frontal (anterior mid-scalp) hair loss.”
The results were quite strong in several regards. Hair growth and cessation of hair loss were noted in significant numbers of patients and to a significant degree. In the vertex group, after two years “66% of men were rated as having improved hair growth…compared with 7% of men receiving placebo.” In terms of stopping hair loss, “Only 17% of men receiving finasteride lost hair based on hair count during the two-year studies, compared with 72% of the men receiving placebo.” Men with frontal hair loss experienced a benefit, but it was not as robust as it was for the men in the vertex group.
The conclusion stated, “Based on hair counts, no further hair loss was observed in 83% of finasteride-treated men with vertex hair loss after 2 years and in 70% of finasteride-treated men with frontal hair loss after 1 year. Based on standardized clinical photography, the chances of mild to moderate visible regrowth are 61% on the vertex (with an additional 5% achieving great visible regrowth) after 2 years and 37% on the frontal area after 1 year.”
It is important to understand that androgenic alopecia is different from many other causes of baldness. Some people can lose all their hair from an autoimmune problem, severe weight loss and dietary deficiencies, cancer treatments, or infections, and regrow hair completely. With MPB, once a section of scalp is completely bald, regrowing hair is almost impossible.
It seems that MPB causes a very specific change during miniaturization and regrowth cannot occur once baldness has occurred. This is discussed below, but this explains why attempts to regrow hair should happen before baldness occurs and why starting early is helpful in preserving hair. For a detailed explanation, read the educational sections below.
Side effects in men using finasteride are generally minimal; nonetheless, some patients develop decrease in semen production, a decrease in libido (which resolved with cessation of the medication), or erectile dysfunction. (More about this below) These generally resolve either while taking the medication or when the medication is stopped. However, there is the possibility that permanent changes can occur in some patients. If sexual side effects occur, such as decreased sexual desire or difficulty gaining or maintaining erections, we recommend stopping the medication immediately.
Finasteride should never be considered for pregnant women, and as far as I can see, there are no benefits in that population that outweigh the risk. The FDA has listed it in pregnancy category X. Finasteride also reduces the androgenic effect of DHT in the prostate, which is why finasteride is used to treat enlarged prostates. In fact, finasteride was first used in the higher 5 mg dose to treat enlarged prostates.
There is a great deal more information concerning finasteride that must be reviewed to help you best understand your hair loss. This information can be found below, following the section on minoxidil.
Please read the full PDR for a full, detailed overview of finasteride.
Use of finasteride:
Who can use finasteride and for what conditions?
Finasteride (1 mg) treats male pattern hair loss (MPB or androgenic alopecia) in men only. It has a proven effect on the crown. Its effectiveness in bitemporal recession has not been established.
In general, daily use for three months or more is necessary before benefit is observed. Continued use is recommended to sustain benefit, which should be re-evaluated periodically. Withdrawal of treatment leads to reversal of effect within 12 months.
Finasteride (5 mg) is used to treat men with symptoms of enlarged prostate to improve symptoms like difficulty urinating, to reduce the risk of getting urinary retention (urine trapped in the bladder), and to reduce the number of men who need surgery of the prostate.
Finasteride at any dose is not approved for the prevention of prostate cancer.
Who cannot take finasteride?
There is a very powerful warning about finasteride and the risk it poses for women who become pregnant or who breastfeed.
__The following warning about women and finasteride cannot be overstated.
PREGNANCY: The PDR states, “Finasteride use is contraindicated in women when they are or may potentially be pregnant. Because of the ability of Type II 5α-reductase inhibitors to inhibit the conversion of testosterone to 5 α-dihydrotestosterone (DHT), finasteride may cause abnormalities of the external genitalia of a male fetus of a pregnant woman who receives finasteride. If this drug is used during pregnancy, or if pregnancy occurs while taking this drug, the pregnant woman should be apprised of the potential hazard to the male fetus.”
The PDR also states, “Women should not handle crushed or broken finasteride tablets when they are pregnant or may potentially be pregnant because of the possibility of absorption of finasteride and the subsequent potential risk to a male fetus. Finasteride tablets are coated and will prevent contact with the active ingredient during normal handling, provided that the tablets have not been broken or crushed.”__
For this reason women should never use finasteride.
The medication has not been approved for use in children.
Children should not use finasteride and it should be kept far from their access (as should all medications).
Also, it should never be used in those allergic to any component of the finasteride tablet or medications in the same class of drug (e.g., dutasteride).
Sexual side effects
Sexual side effects can include a decrease in sexual desire (libido), a decrease in semen volume, or erectile dysfunction (ED). These side effects were noted in 3.8% of men taking finasteride and in 2.1% of men taking a placebo (“sugar pill”). Breaking it down by each side effect, a decreased libido occurred in 1.8% of men (1.3% who took placebo), erectile dysfunction in 1.3% (.7% on placebo), and decreased semen volume in .8% (.4% who took placebo). A very small number of men experience other ejaculation disorders.
The PDR states, “Resolution occurred in men who discontinued therapy with finasteride tablets due to these side effects and in most of those who continued therapy. The incidence of each of the above adverse experiences decreased to <0.3% by the fifth year of treatment with finasteride tablets.”
This all sounds easy and straightforward – the sexual side effects are transient and manageable – but that is not the point. The point is that these side effects can happen. If you are one of the people that notices a change, the statistics about side effects occurring for a small number of patients won’t matter. Whether to continue the medication or not is an intensely personal choice. That is why it is so important to think about what it would mean if you were one of those few men who has an issue. A tiny decrease in semen volume may be trivial, but erectile dysfunction may not be, even if it goes away with time.
Lastly, there is the possibility a side effect can persist after the drug is stopped. It is rare, but possible. And again, “rare” is meaningless if it happens to you. The studies quoted had very few men discontinue medication due to “drug-related sexual adverse experiences,” which was only 1.2% of men compared to .9% of men who took the sugar pill.
Breast changes
There have been rare reports of breast cancer in men. Some studies have shown breast cancer in men taking finasteride (about 1 in a thousand).
The PDR states that in a 7-year study of 18,882 men “there was 1 case of breast cancer in men treated with finasteride tablets, and 1 case of breast cancer in men treated with placebo.”
They noted, “The relationship between long-term use of finasteride and male breast neoplasia is currently unknown.”
Have any side effects been reported since finasteride was first approved?
These are called post marketing reports. Patients have the ability to report possible side effects voluntarily so a relationship to finasteride may not be established.
Some men have reported breast tenderness and enlargement.
There have been reports of “hypersensitivity reactions such as rash, pruritus, urticaria, and angioedema” (including swelling of the lips, tongue, throat, and face).
Sexual dysfunction that continued after discontinuation of treatment has also been noted. Reports include erectile dysfunction, libido disorders, ejaculation disorders, and orgasm disorders.
The PDR notes that testicular pain was reported.
The PDR states, “Treatment with PROSCAR for 24 weeks to evaluate semen parameters in healthy male volunteers revealed no clinically meaningful effects on sperm concentration, mobility, morphology, or pH. A 0.6 mL (22.1%) median decrease in ejaculate volume with a concomitant reduction in total sperm per ejaculate was observed. These parameters remained within the normal range and were reversible upon discontinuation of therapy with an average time to return to baseline of 84 weeks.
The PDR, in referring to post marketing reports, also states, “male infertility and/or poor seminal quality were reported rarely in men taking PROSCAR for the treatment of BPH. Normalization or improvement of poor seminal quality has been reported after discontinuation of finasteride. The independent role of PROSCAR in these events is unknown.”
Yes.
Children: Finasteride cannot be used in children.
Women: Finasteride is category X for a reason. It can lead to birth defects. Finasteride cannot be used in pregnant, possibly pregnant, or nursing women and should not be used in women at all. If a woman who is pregnant, is breastfeeding, or could possibly be pregnant, touches a tablet, she must wash her hands immediately with soap and water and contact her physician.
Elderly: Men aged 65 and over weren’t included in the early FDA studies on baldness, which used the low 1 mg dose of Finasteride. However, the PDR states, “Based on the pharmacokinetics (the way a drug behaves in the body) of finasteride 5 mg, no dosage adjustment is necessary in the elderly.”
Liver impairment: Since finasteride is metabolized in the liver, men with liver impairment may not be able to clear finasteride from the body as easily as men with normal livers.
Kidney impairment: No dosage adjustment is necessary.
Blood donation
Men using finasteride should not donate blood while on the medication or for one month afterward.
Prostate specific antigen
Prostate specific antigen is a blood test used to screen men for prostate cancer. The problem with finasteride is that it can make the PSA level very low in the men who take finasteride—even men with prostate cancer. This fact makes interpreting levels more difficult. A man with a normal level while on finasteride may still have prostate cancer. Interpreting levels must always be done with this knowledge. Every physician involved in your care must know you are on finasteride.
Also, any increases of PSA while on finasteride must be evaluated. The PDR states, “Any confirmed increase from the lowest PSA value while on finasteride tablets may signal the presence of prostate cancer and should be evaluated, even if PSA levels are still within the normal range for men not taking a 5 α-reductase inhibitor (e.g., finasteride).”
Increased risk of high-grade prostate cancer with finasteride and similar drugs
There is the possibility that finasteride may increase the number of men who eventually get high grade prostate cancer. In one study, men aged 55 and over with a normal digital rectal examination and normal PSA “taking finasteride 5 mg/day (5 times the dose of finasteride tablets used for hair loss)…had an increased risk” of higher grade prostate cancer. Some feel that the finasteride makes it easier to detect these higher grade cancers, but it is a risk that should be acknowledged.
The PDR does state, “Whether the effect of 5α-reductase inhibitors (like finasteride) to reduce prostate volume, or study-related factors, impacted the results of these studies has not been established.”
Is it possible to overdose on finasteride?
In clinical studies, single doses of finasteride up to 400x the dose given for hair loss and multiple doses of finasteride up to 80 g/day given for three months did not result in adverse reactions. At the moment, the PDR does not recommend any specific treatment for an overdose, though extremely high doses can kill rats.
Are there any drug interactions?
The PDR states, “No drug interactions of clinical importance have been identified.” Compounds that have been tested include antipyrine, digoxin, propranolol, theophylline, and warfarin and no clinically meaningful interactions were found.
Also, finasteride (1 mg or more), was used in studies with “acetaminophen, acetylsalicylic acid, α-blockers, analgesics, angiotensin-converting enzyme (ACE) inhibitors, anticonvulsants, benzodiazepines, beta blockers, calcium-channel blockers, cardiac nitrates, diuretics, H2 antagonists, HMG-CoA reductase inhibitors, prostaglandin synthetase inhibitors (also referred to as NSAIDs), and quinolone anti-infectives without evidence of clinically significant adverse interactions.”
This lack of effect on other medications and inability of other medications to affect a drug (finasteride) is exceedingly uncommon. Still, interactions that have yet to be identified are possible. Therefore, if any should occur report them to the FDA at 1–800–FDA–1088 or http://www.fda.gov/Safety/MedWatch/default.htm.
What is male pattern baldness (MPB)?
Androgenetic alopecia (AGA), or male pattern baldness (MPB), affects both genders. A receding hairline along the temples “affects 98.6% of men and 64.4% of women.” Hair loss on the very front of the hairline affects nearly 60% of women over the age of 80 years, while 75% of 80-year-old men are bald in both areas.
However, hair loss is not just a condition of the elderly.
From adolescence on, those who are genetically inclined to going bald, start losing hair. In fact, over 60% of men have experienced significant hair loss by 35. 20% of men are losing their hair in two places in their early twenties: the hairline and the crown. Those two areas of hair loss ultimately blend into the fully bald top of the head with which we are all too familiar. The remaining “horseshoe” pattern of hair often is all that remains.
As you will read below, even before baldness begins to appear, the process of shrinkage or “miniaturization” of hair follicles begins—and once it has progressed too far, it is irreversible—or nearly so.
Hereditary factors explain its running in families (mother or father) and it is a predetermined altered biology that makes most people experience hair loss. It was once thought that testosterone was the culprit. People believed being a man meant having lots of testosterone, which meant that baldness had to be accepted as a natural consequence of manhood and aging. As you will read in the next section, our understanding has progressed significantly.
Why do men lose their hair?
For as many as 20% of men, hair loss can begin before their 21st birthday. What is it that makes men so susceptible to male pattern baldness (androgenic alopecia)? Hair loss is due to dihydrotestosterone (DHT), the hormone testosterone becomes when it is exposed to an enzyme called 5 alpha reductase. DHT makes the hair follicles vacate a man’s scalp by slowly destroying clusters of hair follicles. DHT causes the hair follicles to miniaturize over many years. The process may begin slowly but the progression is relentless.
At first, for reasons you will read below, only thinning of hair is noticed, not clear baldness. Some men embrace the process, shaving their heads in response, but many regret the appearance of aging while they still feel young and vital. Fortunately, we have an understanding of how DHT undermines hair growth and therapies exist to interrupt or slow the process.
How does DHT cause hair loss?
DHT causes hair follicles to shrink and lose the ability to create new hairs. It is unknown whether the hair follicles of some men are more predisposed to DHT causing follicles to shrink, or if there are more receptors (places for DHT to “land” on a hair follicle), or if there is some unknown factor but, by blocking DHT, hair loss can be slowed or even reversed in many men.
To follow how this occurs and how finasteride and minoxidil can work (along with how they can cause side effects), an understanding of normal hair growth is necessary.
Understanding hair
First, it is important to know that we are born with all the hair cells we will ever have and there are about 5 million of them on the human body. The scalp will have the densest concentration of these hair cells and the average head will hold about 100,000 hairs, though it varies by hair color. Blondes average 150,000, brown-haired people have 110,000, and redheads have about 90,000 scalp hairs. There will never be more. The good thing is that hair can regenerate and every time a hair falls from your head or is found in a brush, another hair stands ready to take its place. Nevertheless, once a hair follicle becomes too shrunken (miniaturized) by the effects of DHT, it becomes exceedingly unlikely that the hair-producing follicle will return to normal function. Totally bald spots will most often remain so. That is why so many young men are starting finasteride before hair loss has taken too heavy a toll.
Normal growth
Hair growth occurs in three different phases and increases in length by about ½ inch/month or 6 inches/year. There are 100,000 hairs on the scalp and they are never all in the same phase at the same time—otherwise we would shed all our hair at once and go through cycles of full heads of hair and complete baldness.
The 3 phases of hair growth
Anagen: This is when a particular hair grows. The root divides rapidly, as rapidly as any cells in the body, and a hair will stay in this phase of growth for 2 to 6 years depending on the individual. If one person’s growth phase lasts 6 years, they might be able to grow long hair; another person with hair that stays in the growth phase for a mere 2 years will have a shorter maximum length. Most hairs (approximately 85%) are in this phase at any one time.
Catagen: This phase is very short (14 to 21 days) and it here that hair stops growing. The outer sheath of hair attaches to the root of the hair and begins to weaken at its base. About 5% of hairs are in this intermediate phase.
Telogen: This phase lasts for about 3 months and the hair cells are completely at rest. Approximately 10% of hairs are in this phase. Hairs called “club hairs” are now ready to be shed at a rate of about 25–100/day. This is normal.
What is abnormal growth or hair loss?
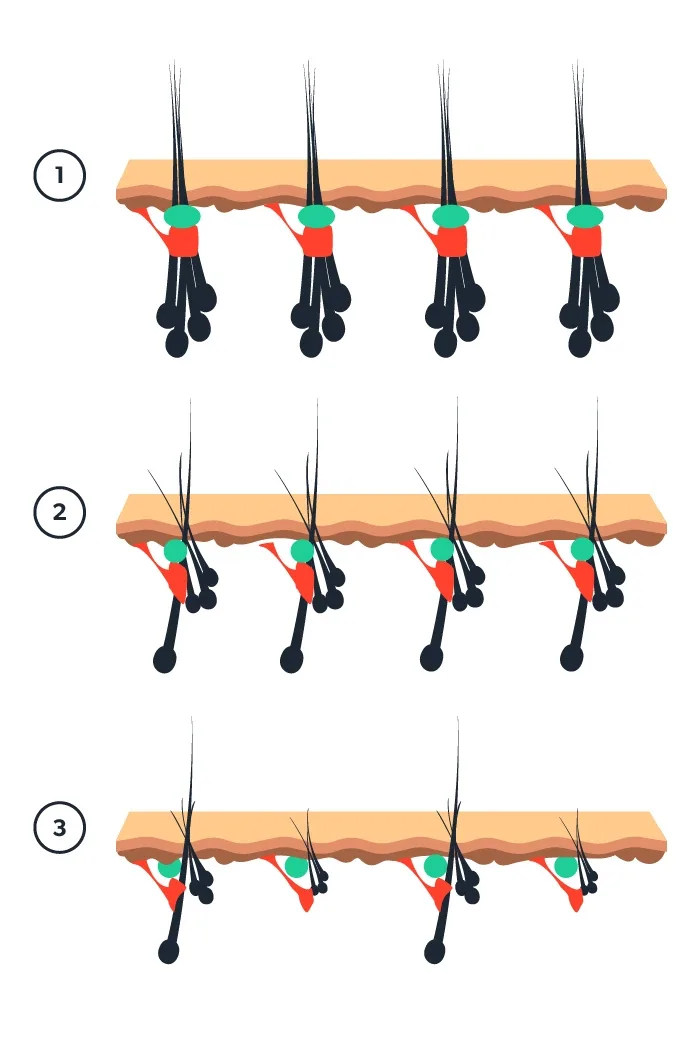
The normal hair follicle is more complex than most people realize. From each hair follicle, multiple hairs sprout. There is a primary hair that goes down all the way to the main root. There are smaller secondary hairs that come out in the same place but don’t go all the way to the root. The primary hair is “grasped” by a tiny muscle called the arrector pili muscle and, as you might have guessed, it is the muscle that makes your hairs “stand on end,” or erect, under different circumstances. Remember that muscle for later. It is important.
As you can see in the drawings, all the smaller hairs, or secondary hairs, are lost before that primary hair gives out (Phases 1–7). That is why so many people will experience thinning hair long before they start to see any true baldness. That is why going bald can sneak up on you.
It may turn out that it is the loss of that arrector pili muscle (when the last hair is lost) that determines whether hair loss is permanent (or nearly so) or can return to normal function. Where the arrector pili muscle “grasps” the primary hair is known as a stem cell niche (a tight little corner where the all-important stem cell can hide), and when that muscle completely separates from the hair and is replaced by fat, hair loss can only be partially restored—at best. In many other conditions that cause complete baldness, the arrector pili muscle stays attached to that last primary hair and complete regrowth is possible.
What this means is that the best time to start to deal with hair loss is before the thinning has progressed too far and before baldness has existed for too many years. To preserve hair, the attempt is best made when the arrector pili muscle is still attached to that last standing, primary hair.
Figure 1: In androgenetic alopecia, shrinkage occurs—initially affecting the smaller secondary hairs. The appearance of “thinning” precedes visible baldness. Only when all of the hairs are affected in an area will baldness be apparent. As shrinkage occurs, the muscle initially loses attachment only to the secondary follicles. When primary follicles eventually lose muscle attachment, the hair loss becomes irreversible. Starting early seems like a good idea. Remember though, no medication is totally safe and even topical medications can have side effects. Potentially, there can be serious side effects of oral finasteride and you need to be aware of them. You must weigh for yourself the importance of the potential benefits of hair maintenance and restoration against the potential for side effects, some of which can be serious.
What are other causes of hair loss?
As you will read, there are many causes of hair loss. While this list is not comprehensive, it gives you a sense of how vulnerable hair growth is and how more than one factor is often responsible.
Genetics
Genes will determine whether you will preserve hair into your 60s or start losing it in your 20s—and the die isn’t cast with a single gene from one parent. A recent study found that there are over 250 genetic areas that influence male pattern baldness (MPB) and they can be inherited from either parent. In the future, it may be possible, by analyzing genetic patterns, to predict who is more likely to develop MPB and even at what stage of life. Moreover, knowing the various genes involved may lead to novel hair regrowth treatments.
Poor health
Anemia, especially iron deficiency anemia is a well-known cause of hair loss. It can occur in women of childbearing years who have heavy periods and do not replace the iron that is lost each month. Men are also susceptible to iron deficiency when eating a diet low in iron. Hair cells divide more rapidly than any others and a balanced diet is essential to healthy hair and nails. One of the problems with identifying hair loss related to diet, stress, or even illness, is that the hair loss can occur months after the triggering event. Pregnant women will often notice hair loss 3 months or more after delivery.
It isn’t just poor health due to iron deficiency or anemia that can be a problem. Vitamin B deficiency, protein deficiency, and even Vitamin A excess can all cause hair loss and be rectified with a well-balanced diet, though the short-term use of vitamins to treat deficiencies may be needed.
Chronic illness
The physical stress (and emotional stress) of any chronic illness can start a cascade of hair loss. Not only stress but chronic illnesses (e.g., cancer, cancer treatment, autoimmune disease) can result in progressive wasting. Inadequate calorie consumption can cause hair loss. It is often seen in anorexia nervosa and eating disorders. Thyroid conditions can alter hair growth dramatically. Too much thyroid hormone and the hair can become thin, too little and it can become coarse and brittle. Uncontrolled diabetes is a cause. Realistically, while some conditions are more frequently associated with shedding hair, any illness can cause hair loss, even something as common as the flu.
Autoimmune disease
Autoimmune diseases like lupus are notorious for causing hair loss occasionally damaging skin so deeply baldness results. Alopecia areata is an autoimmune disease (sometimes brought on by stress) that can cause areas of complete baldness. It is interesting that the arrector pili muscle remains attached to the base of the primary hair in alopecia areata and complete hair regrowth will occur—unlike androgenic alopecia where the connection is ultimately lost and hair regrowth will not occur at that point. Seborrhea and psoriasis are two skin conditions that can occur in multiple locations on the body and can cause hair loss when they affect the scalp.
Scarring from acute injury or chronic inflammation
This is logical. Damage to the skin from any chronic inflammation or from a deep injury like a burn or a deep widespread injury can damage hair follicles. Some autoimmune disease can cause “scarring alopecia” and hair regrowth will not occur. Unlike almost all the other conditions described, hair will not usually regrow over a deep scar.
Infections
Tinea Capitis, which is caused by ringworm, is the most familiar infection people know. It is usually seen in children and causes single or multiple round patches of baldness with broken hairs. The cause is a fungal infection. Sometimes these patches can be difficult to distinguish from seborrhea or psoriasis. Less familiar are the two types of Piedra (Translation—Stone), which is caused by a fungus and can lead to hard nodules on the scalp.
Hormones
Hair growth cycles are very sensitive to hormone levels. Some women may notice hair loss when starting certain hormone-based, birth control methods while others can see acceleration of lost hair when they stop their birth control. Polycystic ovary syndrome (PCOS) causes an excess of anabolic steroids and hair loss is one consequence. Men who use testosterone to bolster their athletic performance (unprescribed) can see male pattern baldness develop rapidly in the same way.
Medications
Medications can either affect the hair growth phase, which is the anagen phase, or they can affect the telogen phase, which is the short resting phase. Baldness due to the first is called anagen effluvium and it is called telogen effluvium if due to the second. Medications like chemotherapy that affect rapidly dividing cells (like hair cells) will cause anagen effluvium and the sudden loss of hair. Other medications cause hair to go into the resting phase and result in telogen effluvium. The names do not matter but the list of medications below and the hair loss they may cause matter a great deal. Note if you are taking any and if they might be playing a role. Other medications may cause hair loss other than those on the list below. If you noticed hair loss after starting a medication, even months later, it is worth considering a link.
Anticoagulants (blood thinners): Hair loss typically begins about 3 months after starting the medication. Drugs include warfarin (Panwarfarin, Sofarin, Coumadin) and heparin injections.
Gout medications: Allopurinol (Lopurin and Zyloprim)
Beta blockers: Beta blockers include Atenolol (Tenormin), Metoprolol (Lopressor), Nadolol (Corgard), Propranolol (Inderal, Inderal LA), Timolol (Blocadren)
Nonsteroidal anti-inflammatories: Ibuprofen (Advil, Motrin)
Angiotensin-converting enzyme inhibitors: These are most often used to control blood pressure. Examples include, Captopril (Capoten), Enalapril (Vasotec), Lisinopril (Zestril, Prinivil).
Acne medication: Vitamin A can lead to hair loss and Accutane is made from Vitamin A
Antidepressants: Amitriptyline (Elavil, Endep), Amoxapine (Asendin), Clomipramine (Anafranil) Desipramine (Norpramin, Pertofrane), Doxepin (Adapin, Sinequan), Fluoxetine (Prozac), Haloperidol (Haldol), Imipramine (Janimine, Tofranil, Tofranil PM), Nortriptyline, (Pamelor, Aventyl), Paroxetine (Paxil), Protriptyline (Vivactil), Sertraline (Zoloft), Trimipramine (Surmontil)
Anti-seizure medications: trimethadione (Tridione) and valproic acid (Depakote).
Stress related hair loss
Stress alone can cause hair loss but as noted above it is often an additional factor when other causes are at work. Some conditions result from stress alone and can resolve when the stress resolves with complete restoration of hair. Alopecia areata is a predominantly autoimmune process but it has long been known that stressful events, anxiety and even depression precede some cases. It is also known that stressful life events (e.g., death of a loved one, illness, or divorce) can lead to hair loss, resolving completely when the stress is relieved.
Lastly, an inability to control the urge to pull repeatedly on hair can lead to hair loss. This is an impulse control disorder called trichotillomania.
Inappropriate grooming
Even a good thing can be overdone. Excessive pressure placed on hair follicles by pulling hairs too tightly as with ponytails of cornrowing can cause hair loss.
Your doctor may also recommend using minoxidil, an over-the-counter topical medication. You can find out more information about minoxidil below and on this drug information page.
How and when should I apply minoxidil?
Part your hair where it is thinning and carefully apply just 1 ml from the dropper that comes with the bottle of Minoxidil. Then, work it into the area of hair loss. Wash your hands immediately afterward. Do not touch other parts of your body.
What no one tells you is that Minoxidil can stain clothes, linens, etc. That means the best time to apply it is not just before going to bed or in the morning just before putting on a hat. Allow it to dry in the morning before putting on a hat. To prevent touching your still wet, medicated scalp while sleeping when minoxidil can be transferred inadvertently, apply it 2 to 4 hours before going to bed.
Don’t worry about missing a dose or two. Just keep going as you would with your next dose. Doubling up doesn’t make hair loss stop any sooner and missing a dose on occasion doesn’t make you lose all you have gained.
If you apply it as directed twice each day, a 60 ml bottle will last one month.
What about my personal hair care routine (e.g., shampooing, conditioning, dyeing)?
Try to use a mild shampoo, etc., and make an effort to develop a routine of washing and conditioning before using Minoxidil—never after.
You can use grooming products after applying minoxidil but the medication should have dried.
You also can have your hair colored or treated but do not apply the last dose of medication on the day you are having your hair treated.
Treatments can irritate the scalp and this can affect the absorption of the medication. Also, for the same reason, do not apply Minoxidil for 24 hours after a treatment as irritation to the scalp might alter absorption.
How quickly will minoxidil work?
Minoxidil 5% used twice/day can work more quickly but 4 months is generally needed to see results. In fact, in the first month very fine hairs, called lanugo hairs, can be shed at an increased rate. They are soon replaced by similar tiny hairs that will be replaced by thicker hairs of your own hair color.
If hair loss has not slowed after a month, it is best to stop the medication and see your doctor. Something else may be contributing to or causing your hair loss.
Also, no medication works on all patients and Minoxidil, again, is not an exception to that rule.
How long will the effects of minoxidil last?
Any hair that is regrown will last only as long as minoxidil is used. Stop Minoxidil and it will fall out usually in 3 or 4 months. Also, hair loss can continue even if at a slower pace while using Minoxidil. In studies, hair regrowth with the continuous use of Minoxidil 5% has not been shown to last beyond 4 years.
What is minoxidil and how does it work?
Minoxidil was first released in the 80’s and, used alone, never proved to be the miracle cure that people anticipated. However, over the years, the use of minoxidil has been shown to slow or stop the progression of hair loss for people suffering from “androgenic alopecia,” which basically is male pattern baldness. The exact mechanism of action in the body is not completely understood. It was initially developed to treat hypertension and, in an early study, hair regrowth was noted in some patients. It tends to work best on younger men who have been experiencing progressive hair loss for fewer than 5 years. In fact, the earlier the better. Minoxidil and finasteride, whether used alone or together, work best when started early. However, even if started early, hair regrowth is more of a secondary gain; stopping hair loss is more likely. Hair regrowth occurs but not in as many men as those for whom it stops or slows hair loss. Interestingly, in the first few months of using minoxidil, loss of fine hairs can occur before they are replaced by thicker, more mature hairs. For some men, it can seem like it is getting a little worse before it improves.
At any rate, in one study (J Am Acad Dermatol 2002; 47: 377–385), men were assigned to three different groups: 5% minoxidil, 2% minoxidil, or placebo. The 5% minoxidil results were superior to 2% minoxidil or placebo. Men using 5% minoxidil had 45% more hair regrowth than the other groups. However, the side effect rate also rose for those men in the 5% group. Women can use minoxidil, but only the 2% solution is recommended for females. The difference between 2% and 5% was not observed in women.
As noted, minoxidil is better at stopping or slowing hair loss than regrowing hair; nevertheless, about 25% of women and 40% of men can experience some regrowth. Also, once the medicine is stopped, the benefit is lost, which means it must be used continuously to maintain the benefit.
Who cannot use minoxidil?
Men must be 18 years old and have androgenic alopecia (male pattern baldness) to use minoxidil 5%. It must be on the top of the head and is ineffective on a receding hairline. If you have patchy hair loss, sudden and abrupt hair loss, and do not know why you have begun to lose hair, do not turn to minoxidil. There are many other causes of baldness (see below) and minoxidil is not recommended in those cases. If you have an itchy, painful, red or inflamed scalp, or a scalp that is in any other way not normal, you may have another reason for hair loss. Give this thought. Minoxidil was developed as a medication for blood pressure. If you are having difficulty with your heart or with your blood pressure (high or low), check with your doctor to be sure it will not be an issue if you use topical minoxidil.
Women cannot use the 5% solution as hair growth in other places is possible. Women should never use minoxidil, at any strength, if pregnant or breastfeeding. Of course, anyone under the age of 18 should not use minoxidil and it should never be applied to children or babies.
It should also be stressed that minoxidil will not work if hair loss is due to any other condition than androgenic alopecia. Please, read the partial list of hair loss causes. Minoxidil would be useless. Moreover, if you have any other causes of hair loss, those should be addressed before attempting to use minoxidil. Other causes of hair loss should be considered even if you have MPB but fail to improve with minoxidil.
Bottom line: do not use minoxidil unless you know you have androgenic alopecia and have ruled out other causes.
Are there any other warnings?
There is the risk of hair growing on other parts of the body if minoxidil is transferred. That is why careful application and handwashing is so important, as well as applying so far in advance of going to bed. If hair growth occurs on other areas, it goes away if the medication is stopped.
It should also be kept out of the eyes and never taken internally. Poison control should be contacted immediately if it is swallowed.
What are some side effects?
Unintended hair growth can occur. Minoxidil can also cause irritation and itching of the scalp where the medication is applied. Women cannot use minoxidil 5%, as wells as pregnant or breastfeeding women No one under 18 can use minoxidil nor should it ever be applied to children or babies.
If you have heart issues, speak with your doctor before using minoxidil. If you have any of the following with use of the medication, stop the medicine go to an emergency room—chest pain, rapid heartbeat, faintness, dizziness, sudden unexplained weight gain, or hand or feet swelling. Consult a doctor if you develop scalp irritation or redness, or unwanted facial hair growth.
Does minoxidil work on my whole scalp?
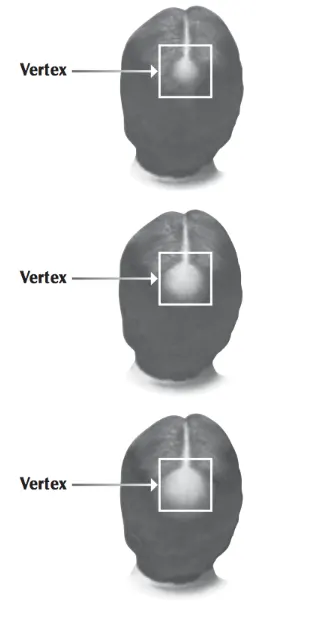
No. Unfortunately, minoxidil does not work on a receding hairline and is approved for use on the “vertex” or the top of the scalp only. (See Photos) Also, once hair loss covers too large an area and persisted for too long, minoxidil is less likely to be beneficial. The younger the patient and the sooner it is started the better the results. That is the case with most conditions and hair loss is no exception.
Can it regrow hair on a completely bald scalp?
No. Male pattern baldness is unlike many other conditions that lead to baldness (e.g., autoimmune diseases, vitamin deficiency, protein deficiency) in that once it has progressed to the point where the scalp is bald, regrowth is virtually impossible. Read the next section to understand why.
Why minoxidil does not work on completely bald spots
The normal hair follicle is more complex than most people realize. From each hair follicle, multiple hairs sprout. There is a primary hair that goes down all the way to the main root. There are smaller secondary hairs that come out in the same place but don’t go all the way to the root. The primary hair is “grasped” by a tiny muscle called the arrector pili muscle and, as you might have guessed, it is the muscle that makes your hairs “stand on end,” or erect. Remember that muscle. It is important.
As you can see in the drawings, all the smaller hairs, or secondary hairs, are lost before that primary hair gives out (Phases 1–7). That is why so many people will experience thinning hair long before they start to see any true baldness. That is why going bald can sneak up on you.
It may turn out that it is the loss of that arrector pili muscle (when the last hair is lost) that determines whether hair loss is permanent (or nearly so) or can return to normal function. Where the arrector pili muscle “grasps” the primary hair is known as a stem cell niche (a tight little corner where the all-important stem cell can hide), and when that muscle completely separates from the hair and is replaced by fat, hair loss can only be partially restored—at best. In many other conditions that cause complete baldness, the arrector pili muscle stays attached to that last primary hair and complete regrowth is possible.
What this means is that the best time to start to deal with male pattern hair loss is before the thinning has progressed too far and before baldness has existed for too many years. To preserve hair, the attempt is best made when the arrector pili muscle is still attached to that last standing, primary hair.
Figure 1: In androgenetic alopecia, shrinkage occurs—initially affecting the smaller secondary hairs. The appearance of “thinning” precedes visible baldness. Only when all of the hairs are affected in an area will baldness be apparent. As shrinkage occurs, the arrector pili muscle initially loses attachment only to the secondary follicles. When primary follicles eventually lose muscle attachment, the hair loss becomes irreversible.
Starting early seems like a good idea. Remember though, no medication is totally safe and even topical medications can have side effects.
Potentially, there can be serious side effects of oral finasteride and you need to be aware of them. You must weigh for yourself the importance of the potential benefits of hair maintenance and restoration against the potential for side effects, some of which can be serious.
Read the full package insert for more information.
Platelet Rich Plasma (PRP)
While the science has not confirmed its utility with clarity, the frequency with which Platelet Rich Plasma being used, either as a stand-alone treatment or in conjunction with hair transplantation, makes it relevant and worth discussing.
Blood contains many components suspended in plasma. Red blood cells, white blood cells, and platelets are the major components. To make PRP, blood is drawn from the patient (when drawn from the patient the resultant PRP is called autologous PRP) and put through two sequential centrifugations. By spinning once to remove the red cells and then again to remove the white cells, a plasma that is rich in platelet cells is created—3 to 7 times as concentrated as in whole blood. Platelets can stimulate many kinds of growth factors and other factors that can enhance hair growth—in theory. PRP has been used in many other areas to accelerate healing and reduce pain. So, the question is, does it work to regrow hair when injected into the scalp of those experiencing androgenic hair loss or hair loss from alopecia areata?
In an article titled “The Effect of Platelet-Rich Plasma in Hair Regrowth: A Randomized Placebo-Controlled Trial” (STEM CELLS TRANSLATIONAL MEDICINE 2015;4:1–7), the effectiveness of using platelet-rich plasma to regrow hair was tested on 20 subjects with androgenic hair loss. A portion of the scalp was injected with autologous PRP and another portion was injected with placebo an equal number of times. The patients then had specialized photography (called a phototrichogram) performed and evaluated by technicians who were blind to what portion of the head received placebo versus PRP. The evaluators were not involved in the treatment. The patients received three courses of therapy 1 month apart and were then followed for two years.
The results were positive in this Italian study. The authors wrote, “At the end of the 3 treatment cycles, the patients presented clinical improvement in the mean number of hairs, with a mean increase of 33.6 hairs in the target area, and a mean increase in total hair density of 45.9 hairs per cm squared compared with baseline values. No side effects were noted during treatment.”
Of significance, four patients had progressive hair loss detected at 16 months and required another course of treatment.
It must be noted that the science to validate the use of PRP has not been performed to the extent it must be to make any definitive statement about its use. The science of its effect has not been completely defined, large numbers of patients have not been tested in multiple double-blind, placebo-controlled, randomized studies, and the follow up to treatment has not been lengthy because of its recent use in the treatment of alopecia.
Laser therapy (LLLT)
The Low Level Laser Device (LLLT) is an FDA approved device. Several devices have been developed that apply low level laser beams to the scalp. The side effect rate is low. The procedure requires the wearing of a cap or use of a comb-like device to apply low level laser therapy (LLLT) to the scalp daily or several times each week. Though few studies have been done, the results have been encouraging if not overwhelming in terms of hair growth. In one study, men and women used LLLT and in both groups hair growth was shown to be statistically significant if slight. The study used real devices compared to sham devices and the photographic results were interpreted by investigators who did not know the origins of the photographs.
Hair regrowth was identified after 26 weeks and patients who had the real therapy “reported overall improvement of hair loss condition and thickness and fullness of hair in self-assessment, compared with sham-treated subjects.”
Hair Transplantation
Hair transplantation is a surgical procedure that moves hair from one place on the scalp to another. While the hairs on the front and top of the head are lost with androgenic alopecia, the hairs on the back and side of the head are preserved, to some degree, even into old age. For that reason, hairs moved from the sides of the scalp to the bald spots of MPB provide a long lasting, if expensive, solution.
There are two major ways to perform the procedure: FUE (Follicular Unit Extraction) and FUT (Follicular Unit Transplantation).
FUE
FUE transplants are the gold standard. Single follicles are removed one unit at a time with a fine surgical tool. These single units of hair are painstakingly placed in tiny cuts that are made in the bald area to reproduce a natural pattern of hair growth. The cuts will also reproduce the normal direction the recipient’s hair would flow.
FUT
With FUT surgery, a strip of hair is shaved from the donor area and then under magnification sliced into tiny donor follicles. Clearly, this isn’t as fine a process. However, there is a great savings in time (and cost), though the recovery can be more prolonged and difficult compared to FUE. Once the hairs are divided, the same process is followed as with FUE, which means the results can be equally refined.